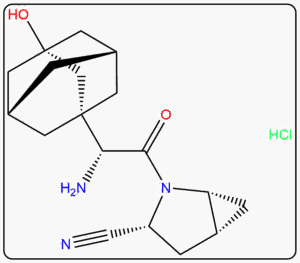
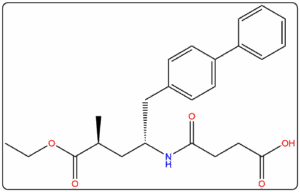
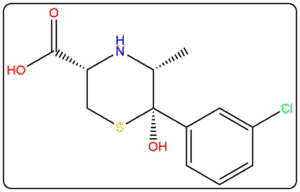
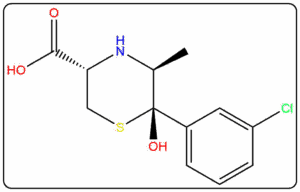
Supplying Pharmaceutical Impurities & Reference Standards Across The Globe Since 2016
×
Nine years of working directly with pharma scientists taught us one thing that a delayed impurity standard or a missing reference material can hold up an entire project.
That is the gap Anjaneey Enterprise was built to close. One delayed impurity standard can push an entire study back by weeks. We have seen it happen, which is why every order we handle comes with real delivery timelines, verified analytical data, and a team that keeps you informed — not one you have to follow up with.
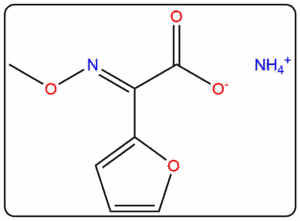
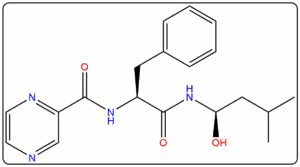
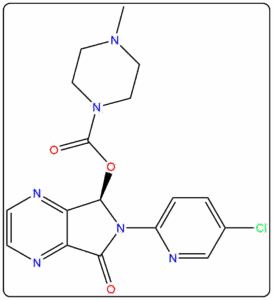
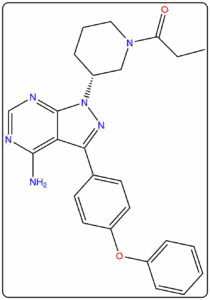
- Reference standards prepared as per IP, EP, USP, and BP pharmacopeial requirements
- Real-time order updates and verified analytical data with every supply
- Cost-optimised sourcing for CROs, pharma manufacturers, and R&D teams